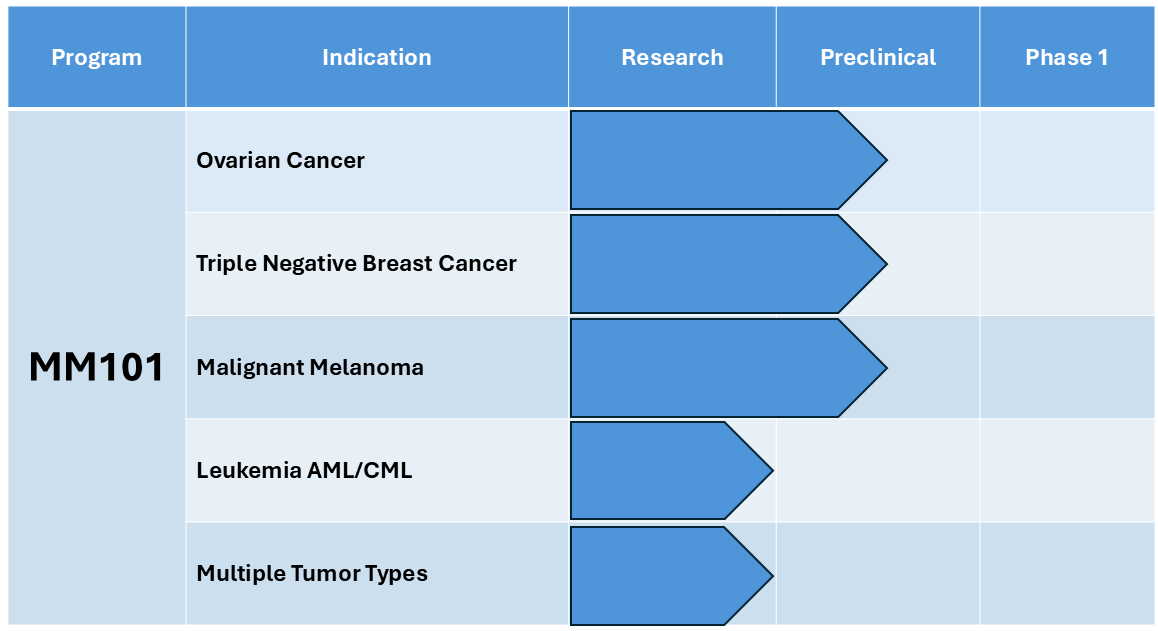
MM101
Lead candidate MM101 is a unique dual-targeting highly-specific peptide that disrupts a key protein, NAF-1 (also called CISD2) required by cancer cells for their survival, thereby inducing cell death. The company has extensive in vitro data in human tumor cells, as well as preclinical data in a wide variety of murine cancer models demonstrating safety and efficacy data.
Unique with regard to most cancer treatments, MM101 exhibits a wide therapeutic window, with little or no toxicity in mice even when given at doses much higher than the effective dose.
We are beginning IND-enabling preclinical studies to advance this drug to human trials. The company intends to evaluate MM101 and additional follow-on candidates as novel treatments for cancer.
MM101’s mechanism of action is unique: no other drugs on the market or in development destroy cancer cells in this way.
MM101 permeates cancer cells, and only cancer cells, due to their different surface membrane characteristics (1, 2). Indeed, we have demonstrated the very strong cancer specificity of MM101 in multiple in vitro and in vivo studies.(1, 3) Once the drug enters the cancer cell, it targets the mitochondria and endoplasmic reticulum and disrupts NAF-1 dimerization, a key regulator of reactive oxygen species (ROS). Cancer cells exhibit high levels of oxidative stress due to their high metabolic rates, which leads to the production of high levels of ROS. MitoMed’s founders have validated NAF-1 as a cancer target, showing that genetically increasing its levels in cancer cells greatly increases tumor growth rate whereas genetically decreasing its level greatly decreases tumor growth (4). By disrupting the NAF-1 dimer it causes the cancer cells to die due to multiple cell death processes that display characteristics of ferroptosis, apoptosis and necroptosis.(1)
These properties strongly differentiate MitoMed’s peptide therapeutics from other cell-penetrating peptide drugs developed in the past.
In nonclinical studies, MM101 is as effective as the cancer therapeutics doxorubicin or taxol, but without their inherent toxicities. MM101 is expected to be active on its own, as well as synergize with many other drugs.
References:
1) Sohn YS, et al. 2022. Chem Sci. 2022 13(23):6929-6941. PMID: 35774163
2) Rowland et al., Cell Death Discov. 2023 Aug 31;9(1):325. PMID: 37652915\
3) Mittler R, et al. 2019 Antioxid Redox Signal. 30(8):1083-1095. PMID: 29463105
4) Darash-Yahana et al. 2016. PNAS 113:10890-5. PMID: 27621439
Mitomed © Copyright. All rights reserved.
We need your consent to load the translations
We use a third-party service to translate the website content that may collect data about your activity. Please review the details in the privacy policy and accept the service to view the translations.
